Essentials of Physical Chemistry by Arun Bahl, B.S. General Chemistry (9th Edition) by Darrell D. 
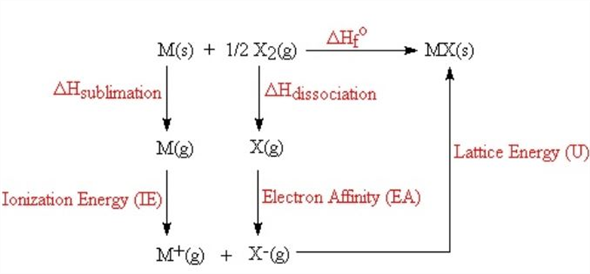
Solid State Chemistry and its Applications by Anthony R West 03. Textbook of Physical Chemistry by Samuel Glasstone 02. Table 4 Representative Calculated Lattice Energies 13 Table 5 Experimental & Theoretical Lattice Energies in Kcal perĠ1. Tables Table 1 Selected Enthalpies of Sublimation at 298 K 9 Table 2 Selected Bond Dissociation Enthalpies at 298 K 10 Table 3 Summary of Reactions in the BornHaber Cycle for the 01 Introduction of Lattice Energy 02 02 Factors Affecting Lattice Energy of a Crystal 03 03 Attraction & Repulsion in a Crystal 04 04 The Relationship between Lattice Energies and PhysicalĠ5 Determination of Lattice Energy 06 06 The Born-Haber Cycle 07 07 Born-Haber Cycle: Explanation with an Examplesīorn-Haber Cycle for Sodium Chloride Born-Haber Cycle for Cesium Fluoride Born-Haber Cycle for Lithium FluorideĠ8 Importance of Born-Haber Cycle 13 09 Applications of Lattice Energy Determination 13 10 Problem 14 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
